
Chem Expt 3 Reacn. of NaCl & K2Cr2O7 (+ H2SO4) Potassium dichromate, Vapor
Reactants: K2Cr2O7 - Potassium dichromate (VI) Other names: Potassium dichromate , Chromium potassium oxide , Dipotassium dichromium heptaoxide. show more Appearance: Red-orange crystalline solid ; Orange-to-red crystals H2O2 - Hydrogen peroxide Other names: Dioxidane , Oxidanyl , Perhydroxic acid. show more

K2Cr2O7 + H2So4 + Feso4 / Используя метод инноэлектронного баланса,расставьте коэф Don't
Balanced Chemical Equation 0 K 2 Cr 2 O 7 + 3 H 2 S + H 2 SO 4 → 0 Cr 2 (SO 4) 3 + 0 K 2 SO 4 + 4 S + 4 H 2 O Warning: Some compounds do not play a role in the reaction and have 0 coefficients. Make sure you have entered the equation properly. Warning: One of the compounds in K2Cr2O7 + H2S + H2SO4 = Cr2 (SO4)3 + K2SO4 + S + H2O is unrecognized.

😊 H2o2 k2cr2o7. Redox uncertainty Cr2O7 + H2SO4 + H2O2? chemhelp. 20190226
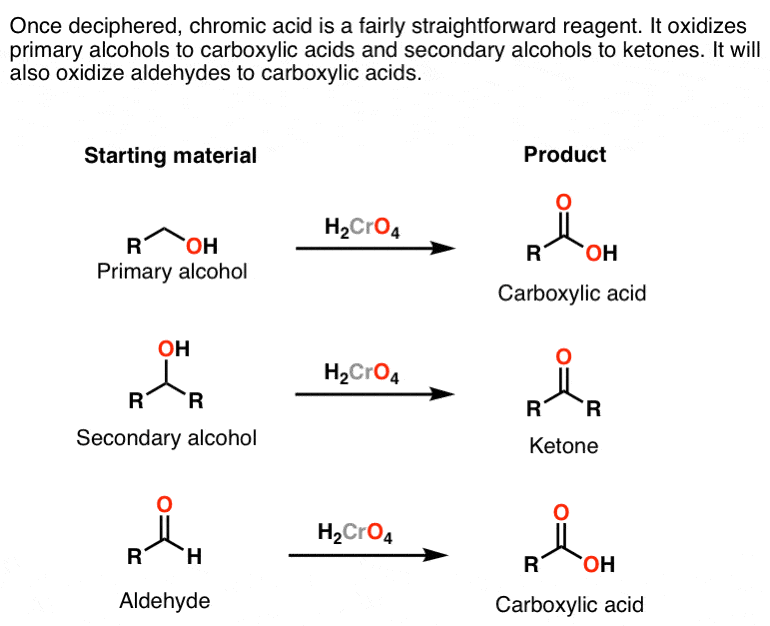
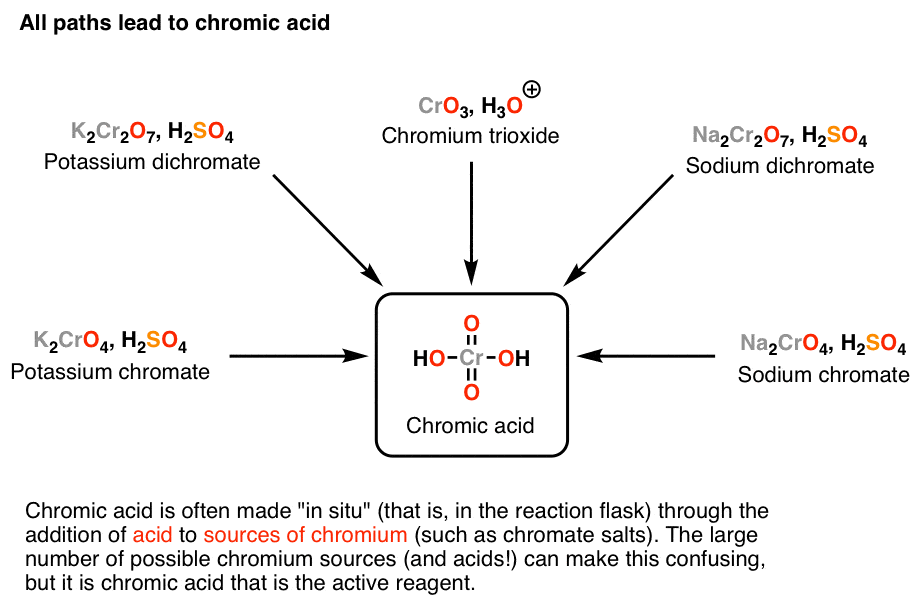
Oxidation by Chromic Acid. One of the reagents that is commonly used for oxidation in organic chemistry is chromic acid. This reagent is straightforward to use once deciphered. However, there are a vast number of different ways that textbooks (and instructors) show it being used in reactions. Chromic acid, H 2 CrO 4, is a strong acid and a.

😊 H2o2 k2cr2o7. Redox uncertainty Cr2O7 + H2SO4 + H2O2? chemhelp. 20190226
James Ashenhurst ( MasterOrganicChemistry.com) 14.6: Oxidation Reactions of Alcohols is shared under a CC BY-NC-SA 4.0 license and was authored, remixed, and/or curated by LibreTexts. Alcohols can be oxidized using acidified sodium or potassium dichromate (VI) solution. This reaction has been used historically as a way of distinguishing between.
Reagent Friday Chromic Acid, H2CrO4 Master Organic Chemistry
Potassium dichromate is usually prepared by the reaction of potassium chloride on sodium dichromate. Alternatively, it can be also obtained from potassium chromate by roasting chromite ore with potassium hydroxide. It is soluble in water and in the dissolution process it ionizes: K 2 Cr 2 O 7 → 2 K + + Cr 2O2− 7 Cr 2O2− 7 + H 2 O ⇌ 2 CrO2−
Solved Draw the major organic product of the reaction shown.
Types of Redox Reactions Question In the given reaction, K2Cr2O7 +XH 2SO4 +Y SO2 → K2SO4 +Cr2(SO4)3 +ZH 2O. Find X, Y and Z. Solution Verified by Toppr SO2 +2H 2O (SO4)2− +4H + +2e−……(1) (Cr2O7)2− +14H + +6e− 2G3+ +7H 2O…(2) Multiplying (1) by 3 and adding (2), 3SO2 +(Cr2O7)2− +2H + 3(SO4)2− +2Cr3+ +H 2O Completing the reaction,

Potassium dichromate (K2Cr2O7) react with sodium sulfite and sulfuric acid K2Cr2O7+H2SO4
By using jones reagent , we get RCHO group ie , an aldehyde. Jones reagent is a relatively mild oxidising agent. Only a strong oxidising ahent such as chromic acid (H2CrO4) could oxidise an alcohol to carboxylic acid. The oxidising order is as follows -. alkanes -> alcohols -> aldehydes -> carboxylic group. 1 comment.

H2so4 H2o Reaction Mechanism Dowload Anime Wallpaper HD
This video is the practical demonstration of the reaction of Acidified Potassium dichromate (k2Cr2O7+H2SO4) with Hydrogen peroxide (H2O2).Precipitation and d.
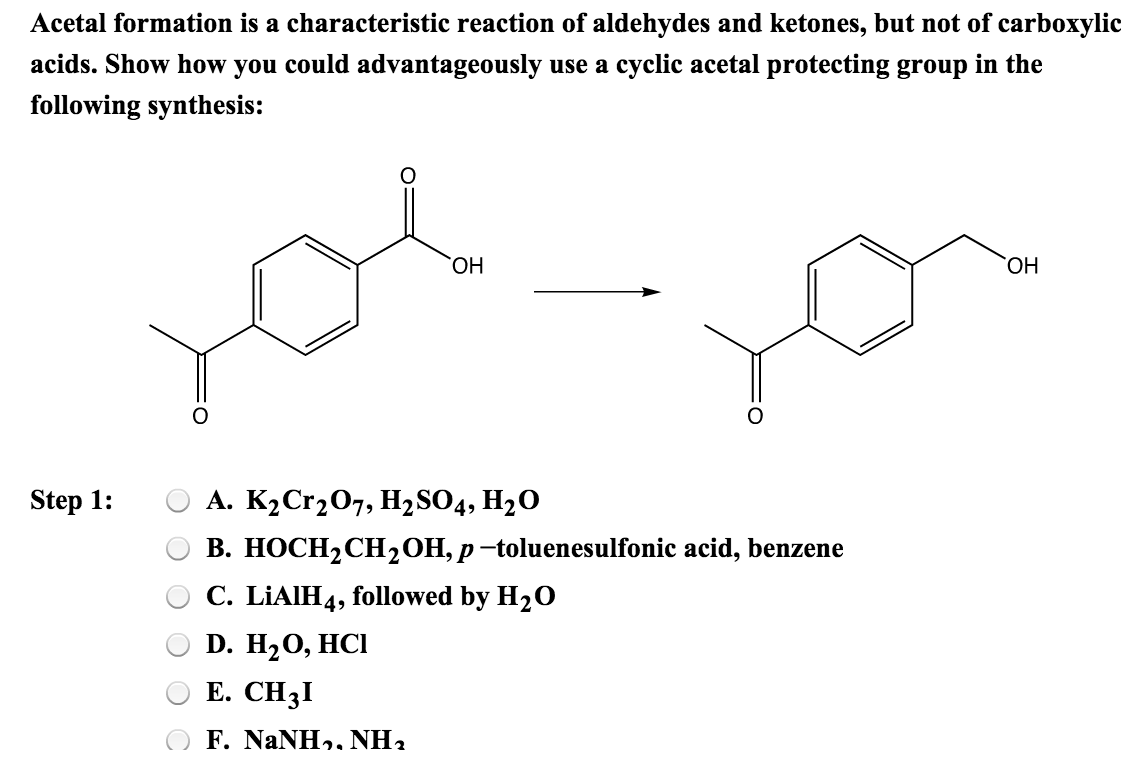
Solved Acetal Formation Is A Characteristic Reaction Of A...
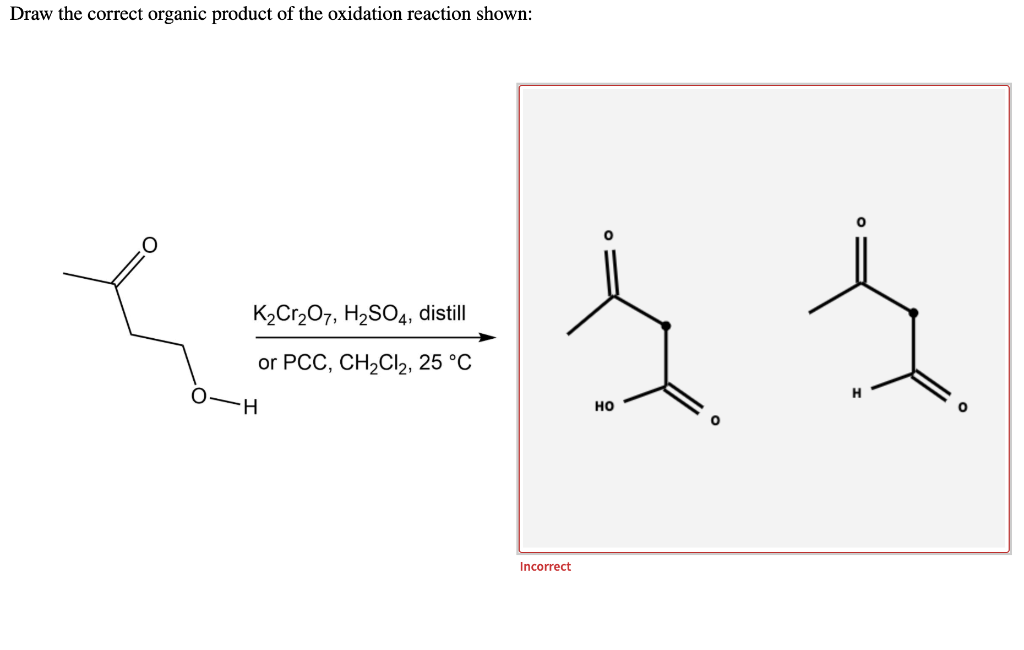
Chemistry Chemistry questions and answers Draw the correct organic product of the oxidation reaction shown: H K2Cr2O7, H2SO4, distill or PCC, CH2Cl2, 25 °C This problem has been solved! You'll get a detailed solution from a subject matter expert that helps you learn core concepts. See Answer

Reaction of k2Cr2O7 with H2SO4 Part 61 class 12 unit 8d,f block elements by Vani ma'am YouTube
The alcohol is heated under reflux with an excess of the oxidizing agent. When the reaction is complete, the carboxylic acid is distilled off. The full equation for the oxidation of ethanol to ethanoic acid is as follows: 3CH3CH2OH + 2Cr2O2−7 + 16H+ → 3CH3COOH + 4Cr3+ + 11H2O (3) (3) 3 C H 3 C H 2 O H + 2 C r 2 O 7 2 − + 16 H + → 3 C H.
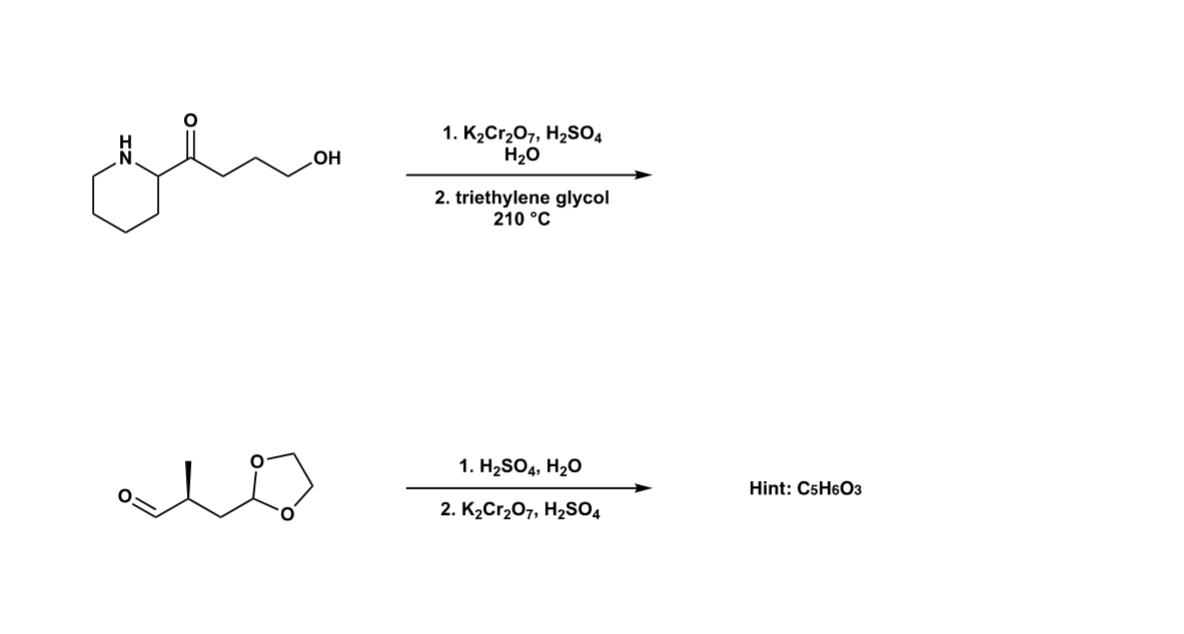
Solved 1. K2Cr2O7, H2SO4 H2O OH 2. triethylene glycol 210 °C
H2SO4 | sulfuric acid | solid + K2CrO4 | | solid = H2O | water | solid + K2Cr2O7 | Potassium dichromate; Potassium bichromate; Dichromic acid dipotassium salt | solid
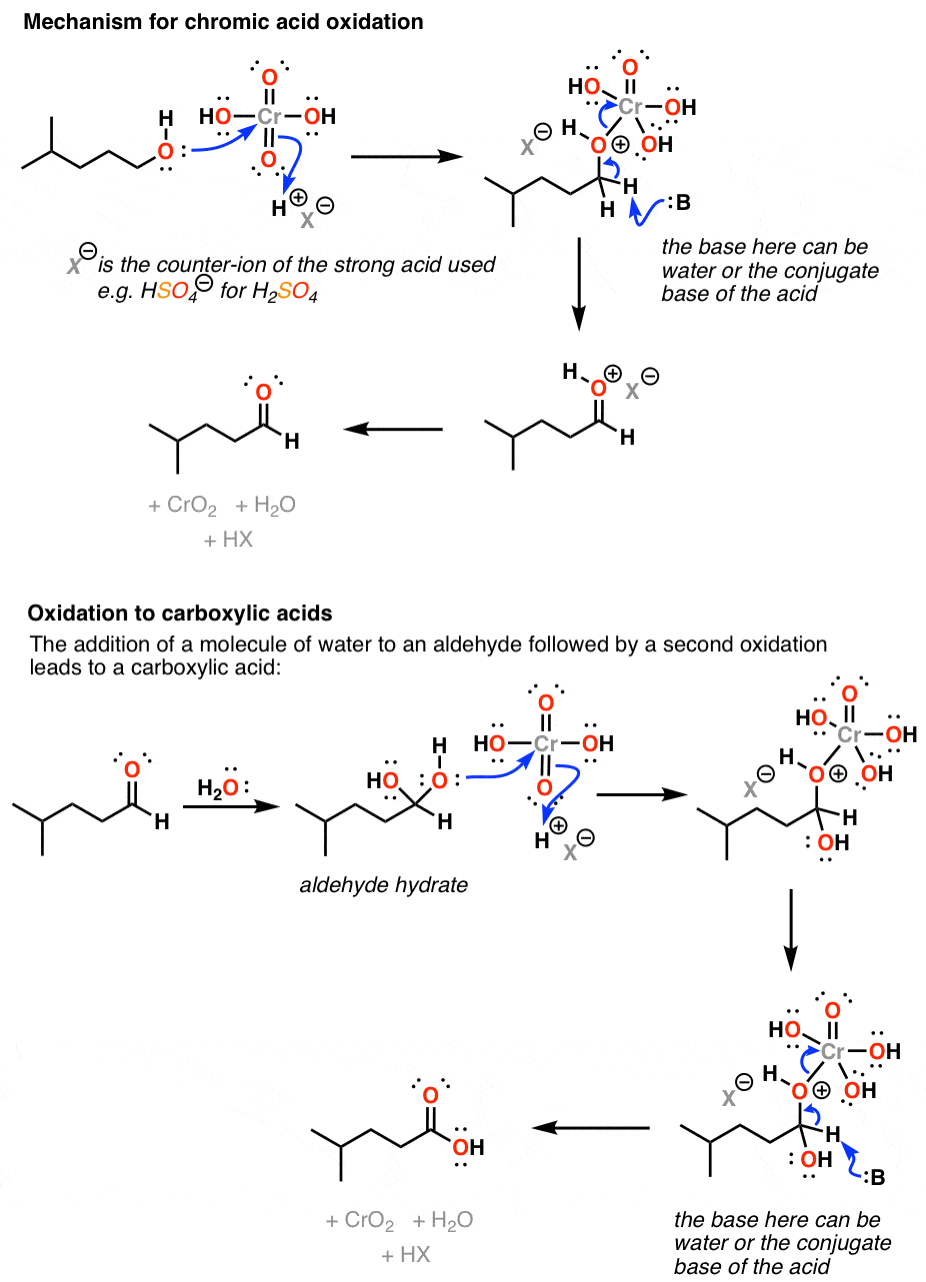
Chromic acid (H2CrO4) as an oxidant in organic chemistry — Master Organic Chemistry
Balanced Chemical Equation 2 K 2 Cr 2 O 7 + 8 H 2 SO 4 → 2 K 2 SO 4 + 2 Cr 2 (SO 4) 3 + 8 H 2 O + 3 O 2 Warning: One of the compounds in K2Cr2O7 + H2SO4 = K2SO4 + Cr2 (SO4)3 + H2O + O2 is unrecognized. Verify 'Cr2 (SO4)3' is entered correctly. ⬇ Scroll down to see reaction info and a step-by-step answer, or balance another equation.
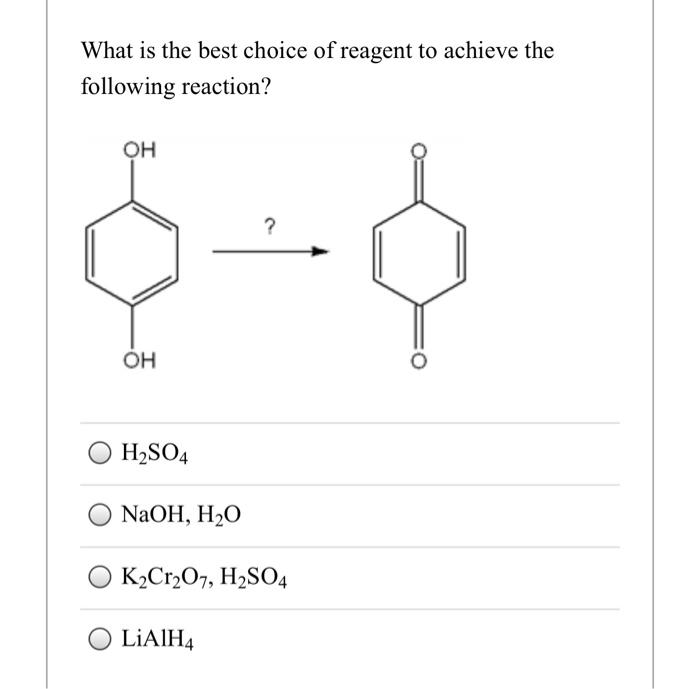
Solved What is the best choice of reagent to achieve the
10 But I thought why not SOX2 S O X 2 While the reaction is stechiometrically correct, it is hard to perform. In excess of HX2S H X 2 S sulfur will be produced. In excess of oxidant sulfate will be produced. And exact balance is impossible to achieve. That said, SOX2 S O X 2 reacts with HX2S H X 2 S finally producing sulfur.
Reagent Friday Chromic Acid, H2CrO4 Master Organic Chemistry
Explanation: The balanced equation is K2Cr2O7 + 3SO2 +H2SO4 → Cr2(SO4)3 +K2SO4 +H2O I think you are referring to the ion-electron method or the half-reaction method. Step 1. Write the skeleton equation The molecular equation is K2Cr2O7 + H2SO4 +SO2 → K2SO4 + Cr2(SO4)3 +H2O
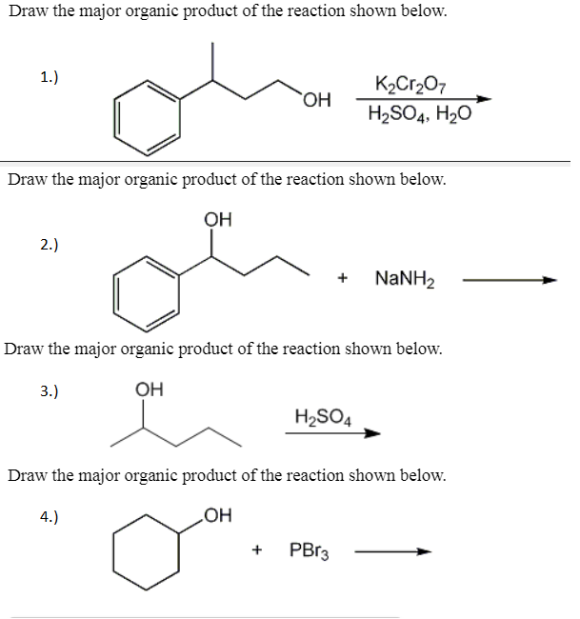
Draw the major organic product of the reaction shown below. K2Cr2O7 H2SO4, H2O WizEdu
Balanced Chemical Equation 303 K 2 Cr 2 O 7 + -5 H 202 + 1212 H 2 SO 4 → 303 K 2 SO 4 + 303 Cr 2 (SO 4) 3 + 707 H 2 O + 707 O 2 Warning: Negative coefficients mean that you should move the corresponding compounds to the opposite side of the reaction.
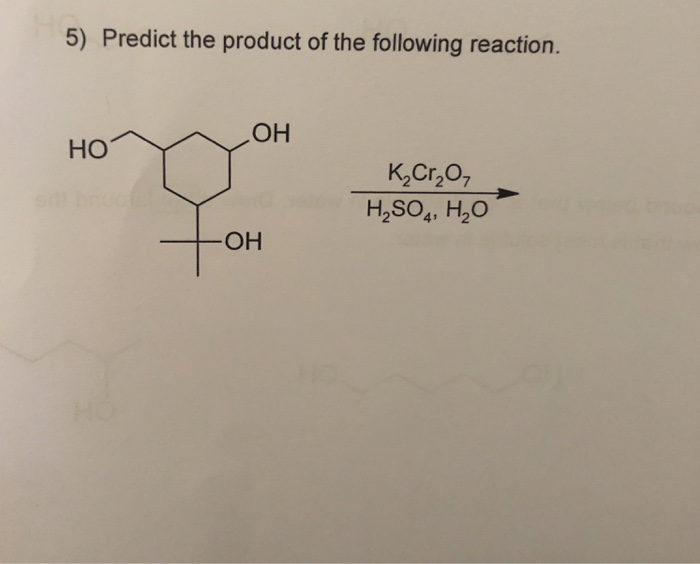
K2cr2o7 H2o Estudiar
Balanced Chemical Equation K 2 Cr 2 O 7 + -1 H 2 O 2 + 4 H 2 SO 4 → Cr 2 (SO 4) 3 + 3 H 2 O + O 2 + K 2 SO 4 Warning: Negative coefficients mean that you should move the corresponding compounds to the opposite side of the reaction. Warning: One of the compounds in K2Cr2O7 + H2O2 + H2SO4 = Cr2 (SO4)3 + H2O + O2 + K2SO4 is unrecognized.